Materials and Methods
Using the available potential vector specimens, we have been able to design a trapping method in order to further investigate the suspected tick and mosquito species. We will focus our efforts in the Orlando area, particularly local nature reserves and hiking trails, given the abundance of cases stemming from people who had visited these areas. Proceeding from this initial trapping, we may sample other suspicious areas around Charleston, SC; Savannah, GA; Tallahassee, FL; and Nueces County, Texas. We will utilize 200 small Sherman live traps baited with seeds and rolled oats, two drag cloths, 65 gravid mosquito traps baited with wet grass clippings or hay and brewer's yeast, and 65 carbon dioxide traps (Saccharomyces cerevisiae).
We will begin by dividing each of the following parks into 2x2 grids stemming from the main trail:
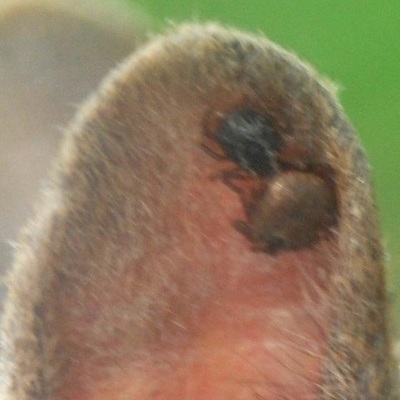
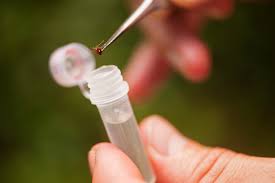
Protocols for tick capture. Possible tick vectors will be collected via small mammal trapping and drag cloth protocols. Small mammals will be captured using Sherman live traps (Figure 1) baited with seeds and rolled oats. We will trap overnight for two nights in each grid at each park. The traps will be set out ~1.5 to 2 hours before sunset. In each grid, set 200 bated Sherman traps in 5 transects of 40 traps per transect (Clark, 2004). The transects will start about 15 meters off from the main trail, and traps will be spaced 15 meters apart. Each transect will be placed 1 kilometer apart from the next. Traps will be collected and each morning and captured small mammals (Figure 2) will be processed. The process of small mammals include: checking for ticks (Figure 3), collecting found ticks (Figure 4), and collecting 2mm ear biopsies from each ear of every captured small mammal. Ticks and ear biopsies will be stored in vials contains 70% ethanol for later laboratory work. The small mammals will then be returned and released into the appropriate transect from which it was collected. The traps will be set out again for another night and the process repeated. After collecting traps and processing captured small mammals on the second morning, the entire trapping process will be replicated in the all grids in every park.
Concurrent drag cloth sampling is necessary to help determine if an established population of tick species is present, especially if mammal trapping
does not yield pertinent information. As per the Centers for Disease Control and Prevention, two tick individuals from different life stages or six tick individuals from the same life stage from the same sampling effort are required to confirm an established population. Drag cloth sampling will be used to capture questing ticks. Drag sampling will be conducted using two 1 meter x 1 meter corduroy drag cloths mounted onto a pole (Figure 5). Collecting ticks via this method requires minimal effort and yields adequate data. The cloth will be dragged along the grass/leaf litter (Figure 6). Drag the cloth in the fields between transects after setting the transect out each evening. Drag the cloth for 15 meters, check the cloth for ticks and collect any ticks found, and then flip the cloth and repeat the process (Falco et al 1992). Continue this process until you have dragged the entirety of the transect from beginning to end to the beginning again.
Later laboratory work will consist of pathology screening, which can be done on small mammal ear biopsies, ticks, and human tissues to determine if the same pathogens are found in all.
Using the available potential vector specimens, we have been able to design a trapping method in order to further investigate the suspected tick and mosquito species. We will focus our efforts in the Orlando area, particularly local nature reserves and hiking trails, given the abundance of cases stemming from people who had visited these areas. Proceeding from this initial trapping, we may sample other suspicious areas around Charleston, SC; Savannah, GA; Tallahassee, FL; and Nueces County, Texas. We will utilize 200 small Sherman live traps baited with seeds and rolled oats, two drag cloths, 65 gravid mosquito traps baited with wet grass clippings or hay and brewer's yeast, and 65 carbon dioxide traps (Saccharomyces cerevisiae).
We will begin by dividing each of the following parks into 2x2 grids stemming from the main trail:
- Bear Creek Nature Trail
- Florida Trail, Mills Creek
- Hal Scott Preserve
- Lake Jesup
- Tibet-Butler Preserve
- Twin Oaks
- Lake Lotus Park
- Historic Babb Landing
Protocols for tick capture. Possible tick vectors will be collected via small mammal trapping and drag cloth protocols. Small mammals will be captured using Sherman live traps (Figure 1) baited with seeds and rolled oats. We will trap overnight for two nights in each grid at each park. The traps will be set out ~1.5 to 2 hours before sunset. In each grid, set 200 bated Sherman traps in 5 transects of 40 traps per transect (Clark, 2004). The transects will start about 15 meters off from the main trail, and traps will be spaced 15 meters apart. Each transect will be placed 1 kilometer apart from the next. Traps will be collected and each morning and captured small mammals (Figure 2) will be processed. The process of small mammals include: checking for ticks (Figure 3), collecting found ticks (Figure 4), and collecting 2mm ear biopsies from each ear of every captured small mammal. Ticks and ear biopsies will be stored in vials contains 70% ethanol for later laboratory work. The small mammals will then be returned and released into the appropriate transect from which it was collected. The traps will be set out again for another night and the process repeated. After collecting traps and processing captured small mammals on the second morning, the entire trapping process will be replicated in the all grids in every park.
Concurrent drag cloth sampling is necessary to help determine if an established population of tick species is present, especially if mammal trapping
does not yield pertinent information. As per the Centers for Disease Control and Prevention, two tick individuals from different life stages or six tick individuals from the same life stage from the same sampling effort are required to confirm an established population. Drag cloth sampling will be used to capture questing ticks. Drag sampling will be conducted using two 1 meter x 1 meter corduroy drag cloths mounted onto a pole (Figure 5). Collecting ticks via this method requires minimal effort and yields adequate data. The cloth will be dragged along the grass/leaf litter (Figure 6). Drag the cloth in the fields between transects after setting the transect out each evening. Drag the cloth for 15 meters, check the cloth for ticks and collect any ticks found, and then flip the cloth and repeat the process (Falco et al 1992). Continue this process until you have dragged the entirety of the transect from beginning to end to the beginning again.
Later laboratory work will consist of pathology screening, which can be done on small mammal ear biopsies, ticks, and human tissues to determine if the same pathogens are found in all.
Protocol for mosquito capture. Possible mosquito vectors will be captured using gravid mosquito traps and CDC-style miniature CO2 bated traps (Holderman et al. 2013) One of each trap will be set the entrance of each park about 15 meters off of the main trail for two days.
Gravid mosquito traps use a fan, fresh grass clippings or hay, a large 32-gallon plastic trash can, 5 grams brewer's yeast, and 30 gallons of tap water is needed. A 1/4 cup of rabbit pellets containing alfalfa and a 2.5-5 gallon plastic container can also be used. The trap is set up by combining the ingredients in the container and leaving it in a sunny location for 5 to 7 days. This creates fermented, grass-infused water. Gravid female mosquitoes are attracted to the water because it provides an organically rich habitat in which to lay their eggs. As gravid females approach the trap to lay their eggs, the fan sucks them into the trap. Mosquitoes trapped by this method are more likely than others to be carrying the pathogen because they have had at least one blood meal (Brown et al 2014). Prepared traps will be deployed one hour before sunset and specimens collected the next morning. These traps are most effective in ecotones (ASTHO, 2005).
We will also employ CDC-style minature CO2 baited traps (Holderman et al 2013). This type of trap mimics the carbon dioxide emitted by the mosquito's natural hosts, and thus lures them in as they attempt to take a blood meal; they are then trapped with a fan (McNelly, 1989). These traps frequently also include a light to further attract mosquitoes. The necessary materials for this trap are a 5 pound block of dry ice, an insulated container (though a drawstring bag may be substituted), and a sealed gel cell battery.
Literature Cited
(ASTHO) The Association of State and Territorial Health Officials. 2005. Public health confronts the mosquito: developing sustainable state and local mosquito control programs. ASTHO, Washington, DC.
Brown, E., J. Fults, G. Hamer, M. Hare, J. Hurley, M. Johnsen, M. Keck, S. Lockwood, M. Merchant, R. M. Sanders, S. Sawlis, T. Sidwa, and S. Swiger. 2014. Texas mosquito management. Texas A&M Agrilife Extension, College Station,TX.
Clark, K. 2004. Borrelia species in host-seeking ticks and small mammals in Northern Florida. J. Clin. Microbiol. 42: 5076-5086.
Falco, R.C., and D. Fish 1992. A comparison of methods for sampling the deer tick, Ixodes dammini, in a Lyme disease endemic area. Exp. Appl. Acarol., 14: 165-173.
Holderman, C., and P. Kaufman. 2013. Spring species composition and sampling techniques for mosquitoes collected within the University of Florida’s natural area teaching laboratory. Proposal, University of Florida, Gainesville, FL.
McNelly, J. R. 1989. The CDC trap as a species monitoring tool. Proceedings, 76th New Jersey Mosquito Control Association, Inc., 26-33.
Brown, E., J. Fults, G. Hamer, M. Hare, J. Hurley, M. Johnsen, M. Keck, S. Lockwood, M. Merchant, R. M. Sanders, S. Sawlis, T. Sidwa, and S. Swiger. 2014. Texas mosquito management. Texas A&M Agrilife Extension, College Station,TX.
Clark, K. 2004. Borrelia species in host-seeking ticks and small mammals in Northern Florida. J. Clin. Microbiol. 42: 5076-5086.
Falco, R.C., and D. Fish 1992. A comparison of methods for sampling the deer tick, Ixodes dammini, in a Lyme disease endemic area. Exp. Appl. Acarol., 14: 165-173.
Holderman, C., and P. Kaufman. 2013. Spring species composition and sampling techniques for mosquitoes collected within the University of Florida’s natural area teaching laboratory. Proposal, University of Florida, Gainesville, FL.
McNelly, J. R. 1989. The CDC trap as a species monitoring tool. Proceedings, 76th New Jersey Mosquito Control Association, Inc., 26-33.